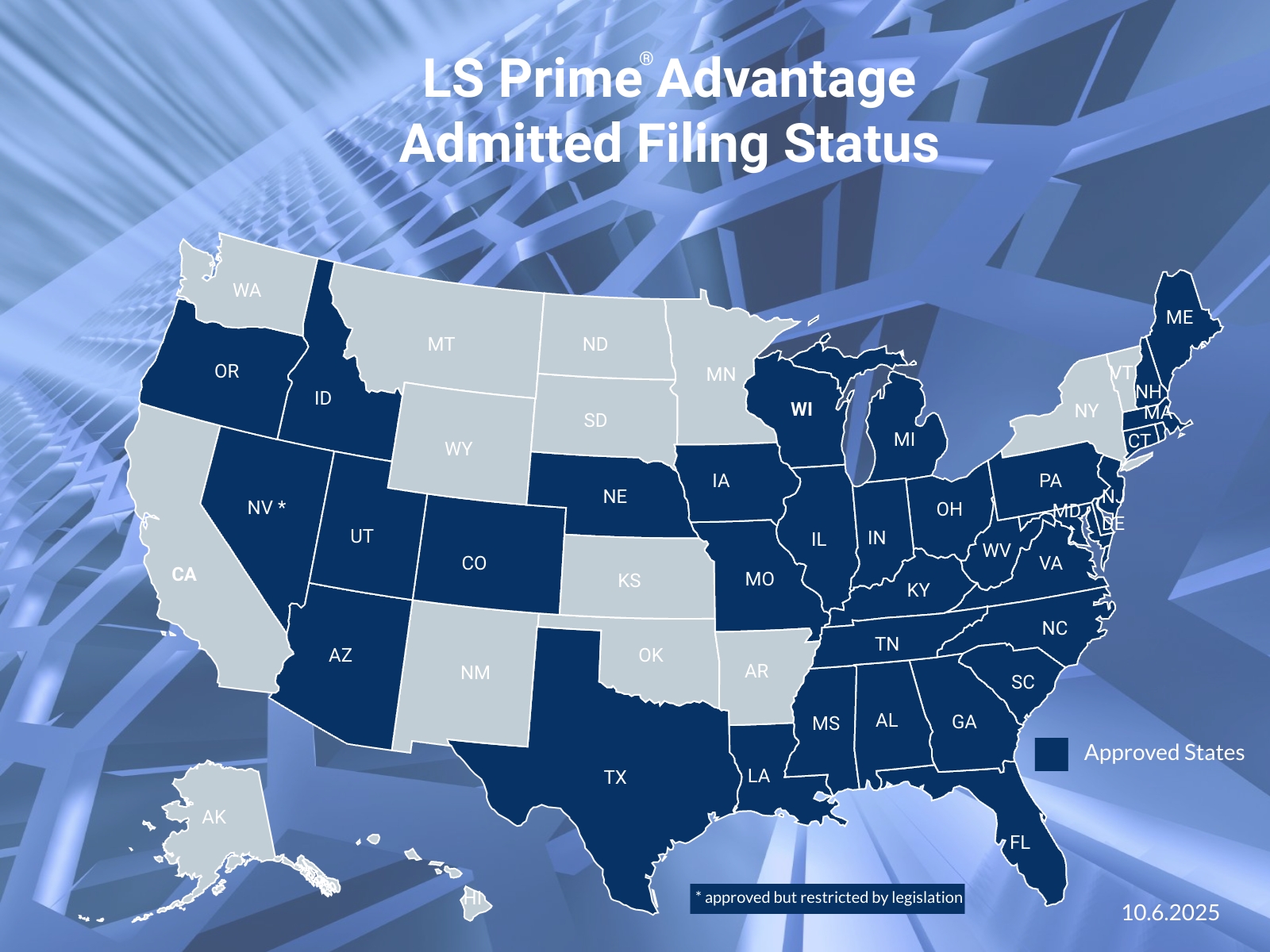
Tailored Life Sciences Insurance
Through appointed retail and wholesale brokers, our industry specialists offer comprehensive life sciences insurance solutions globally.
Feature descriptions are summaries for discussion purposes only and do not modify any policy.
Coverage is determined by the policy terms, conditions, and exclusions as issued and applicable law.


The Latest in Life Science
Injectable Peptides: Out of the Back Alley and Into the Light?
Spanish explorer Ponce de Leon’s “fountain of youth” is legendary. In St. Augustine, Florida, visitors still flock to a tourist attraction named for that mystical spring, hoping that a drink from the city’s natural waters might turn back their biological clocks.
At least the only consequence of indulging in the Ponce de Leon fantasy is a mouthful of sulfur‑smelling water. Today’s pursuit of the “fountain of youth” carries greater risk. Increasing numbers of consumers are injecting themselves with unapproved peptides based on unsubstantiated claims that these compounds bestow regenerative properties. Many of these products are purchased from online sellers operating outside traditional regulatory oversight, where they have not been tested for either efficacy or safety.

Pharmaceuticals Turn to “Niche‑Busters” for Sustained Growth
The pharmaceutical industry is in the middle of a meaningful shift in research strategy. For many years, research teams pursued blockbuster drugs—compounds designed to treat common conditions shared by large segments of the population. Novel treatments for high blood pressure, diabetes, elevated cholesterol, and other widespread ailments were the gold standard.

The FDA AI/ML SaMD Framework: What Companies Need to Know Now
In a recent post on this blog, we examined how artificial intelligence (AI) and machine learning (ML) are reshaping the informed consent process in clinical trials—and why the regulatory and legal frameworks surrounding AI in that context are still catching up. That article focused on one corner of the clinical trial lifecycle. This one broadens the lens. Medical devices, and in particular software as a medical device (SaMD) is a different but related category.